
These properties result from the regular arrangement of the ions in the crystalline lattice and from the strong electrostatic attractive forces between ions with opposite charges. They are not easily deformed, and they melt at relatively high temperatures. Ionic compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. 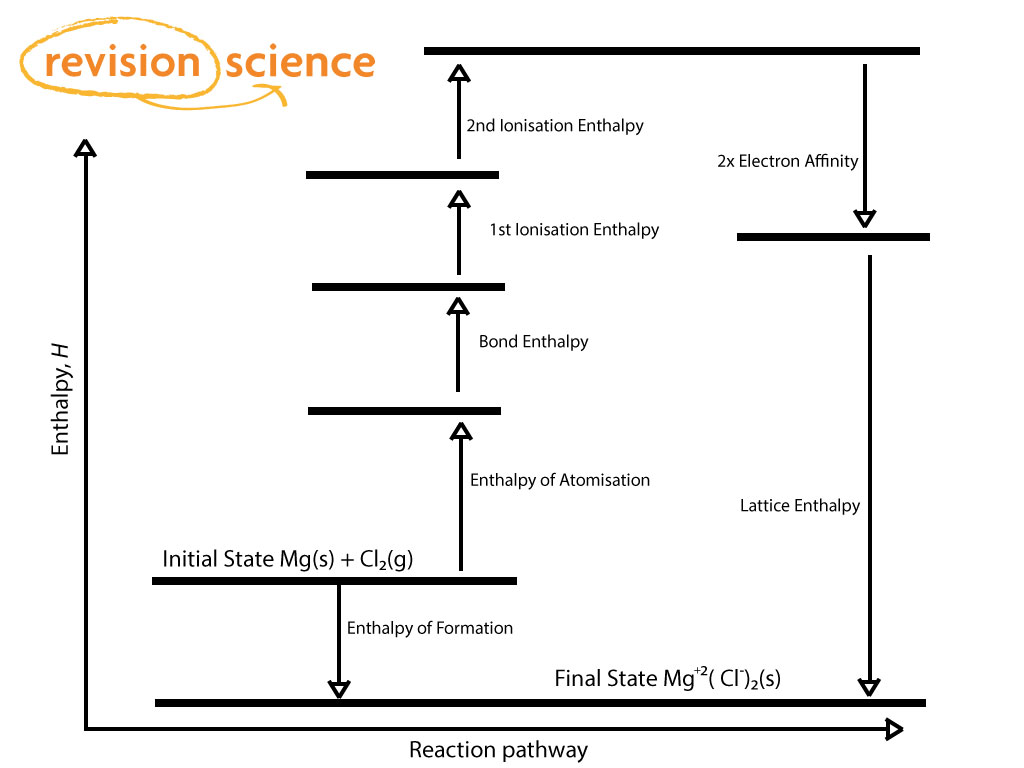
Recall from Chapter 2 "Molecules, Ions, and Chemical Formulas" that the reaction of a metal with a nonmetal usually produces an ionic compound that is, electrons are transferred from the metal (the reductant) to the nonmetal (the oxidant). To use the Born–Haber cycle to calculate lattice energies.To understand the relationship between the lattice energy and physical properties of an ionic compound.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
